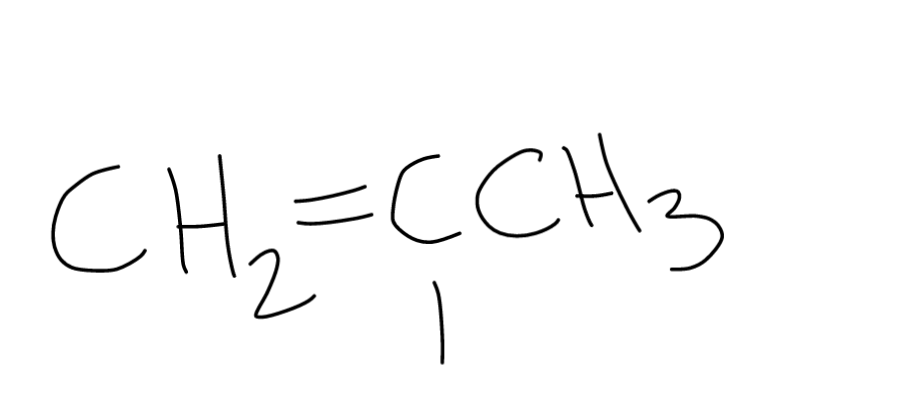Ch2 ch is called vinyl group so when a halogen atom is attached to this group it will be known as vinyl halide and when a halogen atom is attached to benzene ring it is known as aryl halide.
Difference between vinyl and aryl halides.
Alkyl halides and aryl halides are organic halides.
The r group in the structure can be any group with any number of atoms with any type of bonding pattern.
The main difference between alkyl and aryl is that alkyl group has no aromatic ring whereas aryl group has an aromatic ring.
The key difference between these two structural components is the number of carbon and hydrogen atoms.
The term vinyl is often used to describe any alkenyl group.
What is alkyl definition alkyl group alkyl halides 2.
Steric hindrance caused by the benzene ring of the aryl halide prevents s n 2 reactions.
The most important members are the aryl chlorides but the class of.
What is the difference between alkyl and aryl.
R in this case is any other group of atoms.
This conversation is already closed by expert was this answer helpful.
The simplest examples of an aryl halides are bromobenzene or chlorobenzene c6h 5x.
Allyl groups have three carbon atoms and five hydrogen atoms whereas v inyl groups have two carbon atoms and three hydrogen atoms.
Formally this is ethylene h 2c ch 2 with one of the hydrogens substituted by a heteroatom.
Aryl halides are relatively unreactive toward nucleophilic substitution reactions.
Keeping eye on the structural difference of these functional groups we can easily differentiate it between them as allyl group holds three carbon atoms and five hydrogen atoms on the other hand vinyl group has two carbon atoms and three hydrogen atoms.
In high dielectric ionizing solvents such as water dimethyl sulfoxide acetonitrile s n 1 and e1 products may be observed.
In addition the carbon halogen bond is.
In organic chemistry an aryl halide also known as haloarene or halogenoarene is an aromatic compound in which one or more hydrogen atoms directly bonded to an aromatic ring are replaced by a halide the haloarene are different from haloalkanes because they exhibit many differences in methods of preparation and properties.
Vinyl chloride h 2c ch cl is an example.
An aryl halide has general formula c6h 5x in which an halide group x has substituted the aryl ring.
For this reason alkenyl halides with the formula rch chx are sometimes called vinyl halides.
Likewise phenyl cations are unstable thus making s n 1 reactions impossible.
This lack of reactivity is due to several factors.
Rapid s n 2 substitution for 1º halides note there are no β.
What is aryl definition aryl group aryl halides 3.
In organic chemistry a vinyl halide is a compound with the formula ch 2 chx x halide.
Summary alkyl halide vs aryl halide.